energy and place
Essential questions
a. How does energy production and consumption impact place?
b. How does your sense of place, environmental ethic and understanding of our energy needs
influence your perception and decisions relating to energy production and consumption?
a. How does energy production and consumption impact place?
b. How does your sense of place, environmental ethic and understanding of our energy needs
influence your perception and decisions relating to energy production and consumption?
Project Reflection
In this project I learned a lot about energy and energy resources from conducting my own experiment. Throughout this project I learned that the making of diesel fuel while less expensive is less efficient and creates more carbon emissions than gasoline and other alternatives. The knowledge that this project has given me has changed my perspective on how people drive and how it affects the atmosphere. This has taught me that I should be more conservative in my use of cars and fossil fuels.
In this project I learned about the nature of science and how it is conceived by today’s modern minds. I learned a lot about what goes on inside the power plants that are spread across the nation. I also gained a greater understanding of what the process is to find and process the materials used inside of power plants. This has shaped my understanding of the sciences and how scientists are more than just simply blowing things up and looking at the weather.
Scientific Paper
In this project I learned a lot about energy and energy resources from conducting my own experiment. Throughout this project I learned that the making of diesel fuel while less expensive is less efficient and creates more carbon emissions than gasoline and other alternatives. The knowledge that this project has given me has changed my perspective on how people drive and how it affects the atmosphere. This has taught me that I should be more conservative in my use of cars and fossil fuels.
In this project I learned about the nature of science and how it is conceived by today’s modern minds. I learned a lot about what goes on inside the power plants that are spread across the nation. I also gained a greater understanding of what the process is to find and process the materials used inside of power plants. This has shaped my understanding of the sciences and how scientists are more than just simply blowing things up and looking at the weather.
Scientific Paper
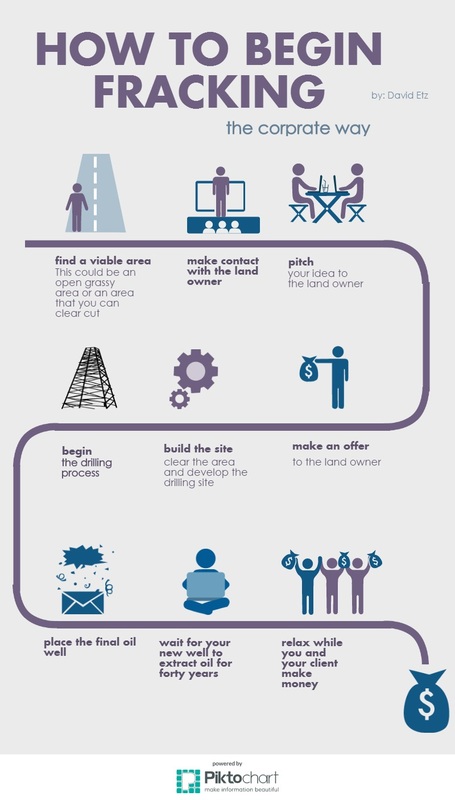
infographic
ELEVATOR pitch for materials project
Hi I’m David Etz and I have a solution for ripped football jerseys. I studied self-healing polymers and how they work. Then, I developed the idea to make certain parts of football jerseys out of the normal material inlaid with self-healing polymers because in high school I have noticed lots of football jerseys tearing under medium stress. I believe that these tears are caused by micro tears that work into the fabric and eventually cause major tears. The self-healing polymers will reconnect the micro tears so that the major tears will be a thing of the past. The self-healing polymers work because inside the mixture there are little catalysts and when those are broken the fluid within spills out into the surrounding micro tear and the reconnects the two torn parts of the fabric. This low cost solution is why you should invest three hundred Osprey Bucks in my product so I can do in depth research and fix the problems of America’s favorite sport.
Self healing football jerseys
December 11, 2014
Nike
Research and design
One Bowerman Drive
Beaverton, OR 97005
Dear Director of Research and Design:
My name is David Etz. I am a junior at Animas High School in Durango, CO and I am doing a project for chemistry in this project we were assigned the task of improving an item of our choosing. In my time playing football at the high school level I have noticed that the football jerseys in high school often rip under medium stress situations and I believe that I have a solution to these problems that could be put into place sometime in the near future when the technology becomes more advanced. This technology is polymers that have the ability to heal themselves, like when you get a cut and your body heals its’ self. The polymers that I am recommending are elastic in nature and could be combined with the elastics of the form fitting jerseys. With the self-healing polymers you would not need to have the entire jersey made of them but simply add the polymers to the areas that are easier to rip such as: the fabric on the collar and the thick areas on the waist line of the jersey and arm holes. These polymers have the self-healing properties in them when you make them. These polymers can be made with over the counter solutions.
The polymers work because they have specific molecules that are made of Ten atoms of Carbon and 12 Atoms of Hydrogen and it is called dicyclopentadiene, This is what is being healed and reconnecting the two sides of the polymer when they talk about self-healing polymers. when torn they release a solution to fill the micro cracks and repair the atomic structure of the material before the problem snow balls and becomes out of hand. This Process would not be able to heal a full fabric tear but it could be able to better prevent the tears in the first place because when the fabric first starts to tear the specially designed molecules release their solution and prevents further damage to the affected area. The University of Illinois completed studies on self-healing polymers and concluded that the bonds when reformed are just as strong and in some cases even stronger than the original molecular bonds. The U of I makes their polymers with simple over the counter solutions that need no special machinery just simply mixing them together. These bonds connecting the polymers are covalent, the scientists at U of I name these polymers polyureas and these polymers are different from other self-healing polymers because they do not require any change in environment or catalysts to begin the healing process, although the healing process increases rate at room temperature for one day, they are completely independent. The heling process can be done because of ring opening metathesis polymerization (ROMP), this allows the polymer to heal because a bond in a closed molecule breaks and then allows the now open molecule to attach to other molecules and creates new bonds to renew the strength of the polymer, in some studies at U of I the healed polymer became fully reattached and broke again in an area which had not been previously healed.
Thank you,
David Etz
Nike
Research and design
One Bowerman Drive
Beaverton, OR 97005
Dear Director of Research and Design:
My name is David Etz. I am a junior at Animas High School in Durango, CO and I am doing a project for chemistry in this project we were assigned the task of improving an item of our choosing. In my time playing football at the high school level I have noticed that the football jerseys in high school often rip under medium stress situations and I believe that I have a solution to these problems that could be put into place sometime in the near future when the technology becomes more advanced. This technology is polymers that have the ability to heal themselves, like when you get a cut and your body heals its’ self. The polymers that I am recommending are elastic in nature and could be combined with the elastics of the form fitting jerseys. With the self-healing polymers you would not need to have the entire jersey made of them but simply add the polymers to the areas that are easier to rip such as: the fabric on the collar and the thick areas on the waist line of the jersey and arm holes. These polymers have the self-healing properties in them when you make them. These polymers can be made with over the counter solutions.
The polymers work because they have specific molecules that are made of Ten atoms of Carbon and 12 Atoms of Hydrogen and it is called dicyclopentadiene, This is what is being healed and reconnecting the two sides of the polymer when they talk about self-healing polymers. when torn they release a solution to fill the micro cracks and repair the atomic structure of the material before the problem snow balls and becomes out of hand. This Process would not be able to heal a full fabric tear but it could be able to better prevent the tears in the first place because when the fabric first starts to tear the specially designed molecules release their solution and prevents further damage to the affected area. The University of Illinois completed studies on self-healing polymers and concluded that the bonds when reformed are just as strong and in some cases even stronger than the original molecular bonds. The U of I makes their polymers with simple over the counter solutions that need no special machinery just simply mixing them together. These bonds connecting the polymers are covalent, the scientists at U of I name these polymers polyureas and these polymers are different from other self-healing polymers because they do not require any change in environment or catalysts to begin the healing process, although the healing process increases rate at room temperature for one day, they are completely independent. The heling process can be done because of ring opening metathesis polymerization (ROMP), this allows the polymer to heal because a bond in a closed molecule breaks and then allows the now open molecule to attach to other molecules and creates new bonds to renew the strength of the polymer, in some studies at U of I the healed polymer became fully reattached and broke again in an area which had not been previously healed.
Thank you,
David Etz
Reflection
The chemistry of materials has shaped our past present and future by allowing us to advance the human body, communication and the rate of imperialistic thinking in the world. The study of different materials has led to a great amount of scientific discoveries such as paper, clothing even insulation that helps to prevent radiation in space. The discovery of even common day things in our lives was once life changing. The discoveries and improvements of these things were all made possible by chemistry and thinking about what physical and chemical properties were best suited for the intended job, whether it is fire resistant or water proof coats. I believe that we will continue to make chemical improvements in all aspects of our lives; we learned that even the youth of the world are still thinking of improvements for the things we use daily.
The structure of matter on the atomic, molecular, microscopic, and macroscopic levels determines the chemical and physical properties because the varying types of bonds and the structure of the molecules creates different strengths, resistances. The bonds create the different strengths because Ionic and Covalent bonds have two different styles of bonding, Ionic has its strength in numbers and can build to make crystalline structures while covalent bonds are none conductive in water because there is not much space in between the water molecules and the covalently bonded molecule. The strength of certain materials varies based on the microscopic arrangement of the same matter such as the graphite in your school pencils but when stripped down to one layer of atoms and built back up in a different way it becomes much stronger.
The structure of matter on the atomic, molecular, microscopic, and macroscopic levels determines the chemical and physical properties because the varying types of bonds and the structure of the molecules creates different strengths, resistances. The bonds create the different strengths because Ionic and Covalent bonds have two different styles of bonding, Ionic has its strength in numbers and can build to make crystalline structures while covalent bonds are none conductive in water because there is not much space in between the water molecules and the covalently bonded molecule. The strength of certain materials varies based on the microscopic arrangement of the same matter such as the graphite in your school pencils but when stripped down to one layer of atoms and built back up in a different way it becomes much stronger.